 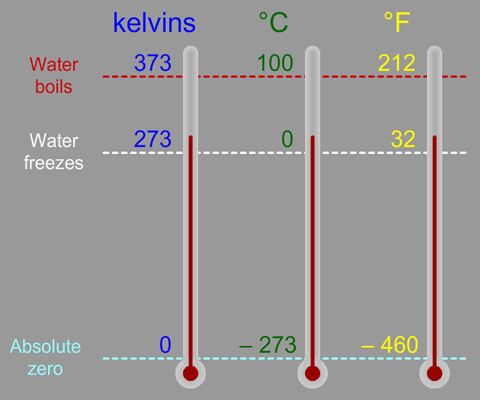
A particle in a gas will move much more than one that is a liquid, which is more than that in a solid. Particles will move differently depending on the state of matter they are in. The average of all the particles in a given system is considered the temperature of the system. The slower it moves, the lower the temperature it has. The faster a particle is moving, the higher the temperature it is considered to have. Some particles move much faster than other particles. The particles will all move randomly, bouncing off against other particles nearby. Kinetic Theory of Matter states that all matter is made up of small particles, atoms, and molecules, which are constantly in motion. To understand why 0 Kelvin is an absolute which can never be crossed, we first have to understand exactly what temperature is, and to do that we need to understand the Kinetic Theory of Matter. In fact, the entire Kelvin scale is based around absolute zero and 0 Kelvin is the very definition of absolute zero, and just as a refresher, Kelvin units are the same size as Celcius units, the only difference being where they put zero. The discussion of absolute zero can be a bit involved, however, the definition is rather straightforward.Ībsolute zero is defined as ?273.15 Celcius, or ?459.67 Farenheight, or more accurately, 0 Kelvin. Learn more about absolute zero and the attempts to get ever close to the impossible temperature on this episode of Everything Everywhere Daily. That is because temperature has an absolute barrier that can never be broken. One of the interesting things about temperature is that no matter how cold you’ve ever been, or how low of a temperature anyone has ever achieved, you can always get colder, if only by a little bit. Density of crude oil, Density of fuel oils, Density of lubricating oil and Density of jet fuel as function of temperature.Apple | Google | Spotify | Amazon | Player.FM | TuneInĬastbox | Podurama | Podcast Republic | RSS | Patreon 
See also other properties of Oxygen at varying temperature and pressure: Dynamic and Kinematic Viscosity and Specific Heat (Heat Capacity), and Thermophysical properties at standard conditions,Īs well as density and specific weight o f acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, pentane, propane, toluene and water. Temperature Choose the actual unit of temperature: ☌ ☏ K °R Choose the actual pressure: 1 bara / 14.5 psia 10 bara / 145 psia 50 bara / 725 psia Specific weight is given as N/m 3 and lb f/ ft 3. The output density is given as kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to estimate the density and specific weight of gaseous oxygen at given temperature and pressure. Oxygen phase diagram Online Oxygen Density Calculator Tabulated values of oxygen density at given temperature and pressure (SI and Imperial units) as well as density units conversion are given below the figures. Where g = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 or 32.17405 Specific weight, γ, has units typically or is defined by the ratio of the weight to the volume of a substance: Density, ρ, has units typically or, and is defined by the ratio of the mass to the volume of a substance: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
